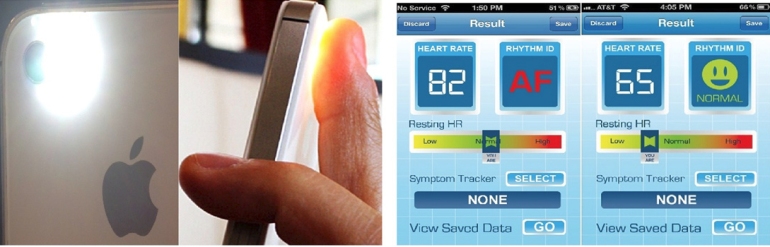
I wrote What is a Pacemaker? Cardiologist’s Guide for Patients and Their Care Providers to fill a gap in available resources for patients who have undergone or are under evaluation for pacemaker implantation. Please consider this a resource for your patients, colleagues, friends, and family.
The fastest growing population segment in the United States, seniors commonly undergo pacemaker implantation. Although doctors’ offices typically provide short pamphlets on pacemaker implantation, there is rarely any comprehensive yet understandable reference material for the patients to obtain…until now. Explaining the “what, why, and how” of pacemaker implantation, this invaluable new guide provides an in-depth summary of pacemakers, from the initial patient evaluation and device implantation to the issues that could potentially arise during a long-term follow up.
Related articles
- When your heart skips a beat (thehindu.com)
- These Pacemakers Adjust To Your Breathing So You Can Stay Active (gizmodo.co.uk)
- Why Stricter Standards Are Needed for Medical Devices (ankinlaw.com)