A recent publication from the National Center for Health Workforce Analysis predicted a shortage of 7080 cardiologists by 2025. Another publication reported recruitment incentives for cardiologists are at “unprecedented levels.” I decided to revisit a paper I published in 2007 (JLW AHHJ 2007) predicting a general cardiology workforce shortage. [1]
I developed this model to project the need for general Cardiologists from 2005-2050 using Matlab (Mathworks, Inc., Natick, MA). The growth in need for General Cardiologists was estimated by incorporating the effect of retirement, prevalence of heart disease, and patient per physician load. At the peak demand in the year 2038, the model projected a need for 62,452 General Cardiologists. Current training durations would result in 29043 General Cardiologists and Fast-Tracking (e.g., third year of Internal Medicine training counted as first year of cardiology) would result in 32533 General Cardiologists. There was evidence of an impending shortage of General Cardiologists that will peak in 2038 resulting in only 46.5% of the projected need for General Cardiologists. This may result from a complex cascade of declining US medical graduates and those matching in Internal Medicine residencies, combined with an increasingly complex cardiovascular disease patient requiring the care of multiple, distinct cardiovascular specialists.
Baseline Data for Modeling the Cardiologist Workforce:
The 35th Bethesda Conference [2] revealed that only 120 of 173 Clinical Cardiac Electrophysiology (EP) spots and 229 of 269 Interventional spots are filled per year. These baseline partial fill rates were used to assess the effect completely filling these subspecialty fellowship positions would have on overall number of General Cardiologists. In 2001, there were 2160 total trainees and 709 first year fellows. In the baseline conditions of the model, the number of first year fellows was taken as 709, 2nd year fellows numbered 726, and 3rd year fellows numbered 725.
There are an estimated 6 cardiologists per 100,000 U.S. residents. This was used as the basis for calculating the number of cardiologists in the US at 16800 in 2005.
Determining Growth in Need for General Cardiologists:
Effect of Retirement: I estimated that 10% of Cardiologists would retire by 2015. Thus, the model uses 1%/year increase in need due to retirement.
Effect of Prevalence of Heart Disease (HD): Heart disease deaths indicate a need for cardiologists however, prevalence of HD is more important than death in determining workforce requirements. In this model, it was assumed the prevalence of HD will grow by 1.7%/year until 2030. As the baby boomer population passes away, the prevalence of HD will decrease by 0.58%/year from 2030-2040 and 0.39%/year from 2040-2050.
Effect of Decreasing Physician-Patient Load: The average physician’s patient load in cardiovascular medicine declined by over a third from 1980-1995. There are a higher proportion of patients who require the care of more than one cardiovascular specialist (e.g., a General Cardiologist, Electrophysiologist, Interventionalist, and/or Heart Failure Specialist). For every 10% decrease in average patient load, 20% more physicians are required. The model uses 2%/year increase in demand due to decreasing physician-patient load.
Effect of Cardiovascular Subspecialty “Fast-Tracking”:
1.The 8th Working Group of the 35th Bethesda Conference suggested a means to allow a 5-year short-track to train general cardiologists. The trainee would complete 2 years of general internal medicine then 3 years of cardiology. This short-track would increase the number of general cardiologists, free up more money for additional trainees, and permit trainees to begin paying on student loans. However, they did not discuss the possibility of “Fast-Tracking” for Interventional or Electrophysiology Fellowships for those who have already completed a 3-year Internal Medicine residency.
2. In this model, “Fast-tracking” would comprise 2 years of a General Cardiology Fellowship then 2 years of either Interventional or Electrophysiology training. This concept of “Fast-Tracking” was incorporated into the model to assess its effect on the General Cardiology Workforce numbers.
Growth in Need for General Cardiologists from 2005-2050 :
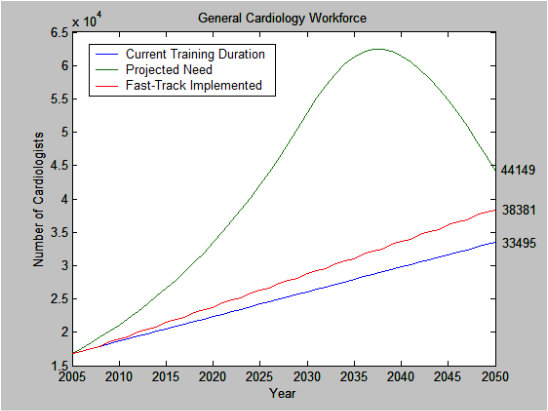
Projections of Cardiology Workforce from 2005-2050. This model incorporates a decrease in heart disease prevalence by 0.58%/year from 2030-2040 and 0.39%/year from 2040-2050. Current training duration (blue line) and Fast-Track (red line) would still result in deficit when compared with model projection of need for General Cardiologists (green line). At the peak demand in the year 2038, there is a projected need for 62,452 General Cardiologists. Current training durations would results in 46.5% (n=29043 total cardiologists) the projected need and Fast-Tracking would result in 52.1% the projected need for General Cardiologists (n=32533).
Current training duration (blue line) and Fast-Track (red line) would still result in deficit when compared with model projection of need for General Cardiologists (green line) from 2005-2050. See Figure. At the peak demand in the year 2038, current training durations and Fast-Tracking would result in 46.5% (n=29043) and 52.1% (n=32533) the projected need for General Cardiologists. Doubling the number of General Fellows trained and incorporating Fast-Track for EP and Interventional fellows would help offset the predicted shortage in General Cardiologists (green line) by year 2020. However, this would result in an oversupply in General Cardiologists by the year 2050.
How accurate was my model predicting the cardiology workforce as of 2015?
The Association of American Medical Colleges (AAMC) publishes a biannual report on the most current data available about active physicians and physicians in training. [3] The AAMC estimated 22038 active cardiologists whereas my model predicted 20515 active cardiologists in 2015. Certainly, care delivery models and market forces (e.g., 2008-2009 recession leading to decreased retirement rates) have affected the cardiology workforce projections. My estimates of the prevalence of heart disease continue to be accurate. In 2012, The Trust for America’s Health [4] found that at present growth rates “the number of new cases of type 2 diabetes, coronary heart disease and stroke, hypertension and arthritis could increase 10 times between 2010 and 2020—and double again by 2030.” Finally, it is difficult to get an accurate trend in cardiologist patient loads over time. The recent Medscape Cardiologist Compensation Report 2016 [5] reported that only a quarter of cardiologists have seen an influx of patients due to the Affordable Care Act (ACA) while three-quarters have not.
Is there an impending shortage of general cardiologists? I suspect my model is accurate though has overestimated the shortage of general cardiologists we can expect moving forward. Several reasons included delayed retirement of current cardiologists, the move towards primary-care directed delivery, and the use of physician extenders. That being said, there have been a host of studies examining the cardiology workforce using a variety of analyses that all point to a shortage of cardiologists moving forward.
It is clearly difficult to place a number on any “shortage” of cardiologists however, there is a common theme to the various studies looking at the future cardiology workforce. The most recent study [6] reported a shortage of 7080 cardiologists by the year 2025. “Projections were developed using the Health Resources and Services Administration’s (HRSA) Health Workforce Simulation Model (HWSM), an integrated microsimulation model that estimates current and future supply and demand for health care workers in multiple professions and care settings.” The 2009 American College of Cardiology Board of Trustees Workforce Task Force [7] reported a deficit of 16000 cardiologists by 2025 and best case scenario of 8000 cardiologist deficit with pointed interventions. The CV workforce model I developed estimated a deficit of 17865 general cardiologists by the year 2025.
Summary:
It continues to be reasonable to estimate a general cardiologist shortage by the year 2025. There is consensus of a likely deficit in the general cardiologist workforce using a disparate set of studies including those looking at practice-level demand, prevalence of CV disease, and implications from recruitment incentives for general cardiologists. Regional variation is to be expected but health systems should be cognizant of the likelihood of a general cardiologist shortage in the near- to mid-term.
References:
1 Williams JL, “Projecting the General Cardiology Workforce Shortage,” American Heart Hospital Journal, V. 5 (Fall 2007), pp. 203-209.
2 Fye WB, Hirshfeld JW. Cardiology’s workforce crisis: a pragmatic approach. Presented at the 35th Bethesda Conference, Bethesda, Maryland, October 17–18, 2003. J Am Coll Cardiol 2004;44:215–75.
3 American Association of Medical Colleges, “2016 Physician Specialty Data Report,” Accessed online at https://www.aamc.org/data/workforce/reports/457712/2016-specialty-databook.html.
4 ”F as in Fat: How Obesity Threatens America’s Future”; Trust for America’s Health Issue Report, Sept 2012; Robert Wood Johnson Foundation. Accessed at http://healthyamericans.org/report/100/.
5 Peckham C, “Medscape Cardiologist Compensation Report 2016,” April 1, 2016. Accessed at: https://www.medscape.com/features/slideshow/compensation/2016/cardiology#page=15
6 U.S. Department of Health and Human Services Health Resources and Services Administration Bureau of Health Workforce National Center for Health Workforce Analysis, “National and Regional Projections of Supply and Demand for Internal Medicine Subspecialty Practitioners: 2013-2025,” December 2016. Accessed online at https://bhw.hrsa.gov/sites/default/files/bhw/health-workforce-analysis/research/projections/internal-medicine-subspecialty-report.pdf.
7 Rodgers GP, Conti JB, Feinstein JA, Griffin BP, Kennett JD, Shah S, Walsh MN, Williams ES, Williams JL. ACC 2009 survey results and recommendations: addressing the cardiology workforce crisis: a report of the ACC Board of Trustees Workforce Task Force. J Am Coll Cardiol 2009;54:1195–208.